How Isothermal Titration Calorimetry (ITC) Measures Biomolecular Interactions

Isothermal Titration Calorimetry has become more prevalent in biomolecular science, research and engineering. With an increasing need to characterise biomolecules and their interactions, it’s important for researchers and scientists across many fields to understand how this unique instrumentation functions.
In this article you’ll discover exactly how ITC instruments can be used to measure biomolecular interactions and how they are used in drug development and other industries.
What is Isothermal Titration Calorimetry?
ITC works by directly measuring the heat that is either released or absorbed during a biomolecular binding event.
Used in quantitative studies of biomolecular interactions, Isothermal Titration Calorimetry is the only known technique that can determine all binding parameters simultaneously in a single experiment. This fact alone makes Isothermal Titration Calorimetry an important component in biomolecular study.
ITC can also measure the affinity of binding partners in their native states, doing so without modification with fluorescent tags or immobilisation.
Common uses of Isothermal Titration Calorimetry
Traditionally, ITC is most commonly used for investigations into protein interactions, including:
- Protein-protein interactions
- Protein-DNA/RNA interactions
- Protein-small molecule interactions
- Enzyme kinetics
In the above interactions, Isothermal Titration Calorimetry provides the most direct route to a more complete thermodynamic characterisation of protein interactions.
Furthermore, over the past few years ITC has seen rapid development and usage uptake. It has gone from from a specialised method focused on understanding the interactions of molecular biological processes to newer applications in protein folding and misfolding. This works to expand the use case of ITC methodology and instrumentation.
What does ITC measure?
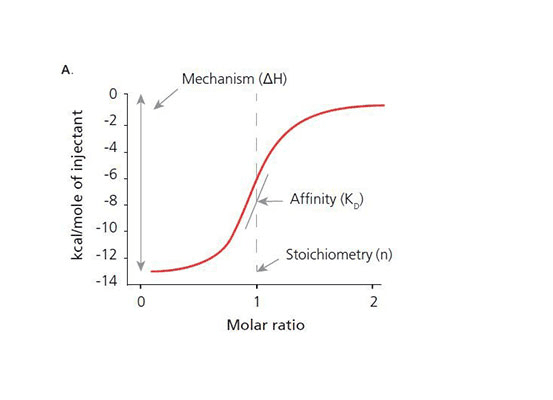
Isothermal Titration Calorimetry measures heat released or absorbed by complexes formed between molecules. This provides information on the affinity of binding partners in a native state. From here, operators can utilise experiment data to generate accurate information on:
- Binding constants (KD)
- Reaction stoichiometry (n)
- Enthalpy (ΔH)
- Entropy (ΔS)
This occurs in part through the introduction of a ligand into the biomolecular sample.
During a binding occurrence, heat will either be absorbed or released. Progressive titration of the ligand into a sample cell containing a biomolecule allows the sensitive calorimeter to take a measurement.
What makes Isothermal Titration Calorimetry unique?
A complete characterisation of epigenetic proteins requires a clear interaction between a specific module in isolation and its mark. Without this interaction it’s difficult to characterise the structure, function and activity of an epigenetic proteins.
What makes ITC unique is that it is the only known method for characterising all known binding parameters in a single experiment. It is therefore more efficient at producing results than most of the other instruments available in the field.
How ITC instrumentation works

The Malvern MicroCal PEAQ ITC is designed for ease-of-use with exceptional sensitivity and works along the following guidelines.
Preparing the thermal core
The microcalorimeter has two cells, one containing water, which is the reference cell. The other cell contains the sample you are testing. The calorimeter keeps these two cells at the same temperature while the heat sensing component detects any temperature difference between each of the cells when binding occurs.
This thermal core is at the heart of every Isothermal Titration Calorimetry experiment. Careful preparation according to the instrument manual is essential for a successful and accurate experiment.
Taking measurements
As stated above, ITC requires progressive titration of a ligand into the sample cell.
The ligand, which is loaded into a syringe, is inserted into the sample cell. From here, aliquots of ligand are injected into the protein sample.
If the ligand binds to the protein it will cause heat changes within the sample. The ITC instrument’s capability means even if the change is only a few millionths of a degree, it can be detected and measured.
In all cases, the quantity of heat measured is directly proportional to the amount of binding.
Analysis of data and results
The molar ratio between the ligand and protein is gradually increased through a series of ligand injections.
This causes the protein to become saturated and less binding occurs between it and the ligand. Heat exchange decreases until an excess of ligand brings about the reaction towards saturation. Once this occurs, the experiment component ends.
From here, data from the instrument can be accessed using software to produce graphs and reports.
ITC and drug development
ITC is predominantly used in drug development specific to the following areas:
- Quantification of binding affinity
- Selection and optimisation of candidates
- Thermodynamic and concentration measurements
- Mechanism of action characterisation
- To confirm intended binding targets in molecules
- Binding specificity and stoichiometry determination
- Enzyme kinetic measurements
With an ever increasing focus on biomolecular research and drug delivery, many research facilities find themselves in search of the right Isometric Titration Calorimetry instrument.
Introducing the Malvern MicroCal PEAQ ITC
When it comes to stand out ITC instrumentation, the Malvern MicroCal is an exceptional frontrunner. Delivering an accurate and sensitive analysis with minimal resource consumption, this is the ideal device for most laboratories. Discover more about this automated solution here. Or contact us today for a full quote.




 02 9541 3500
02 9541 3500