Efficient Production Process for Nanocrystalline CeO2
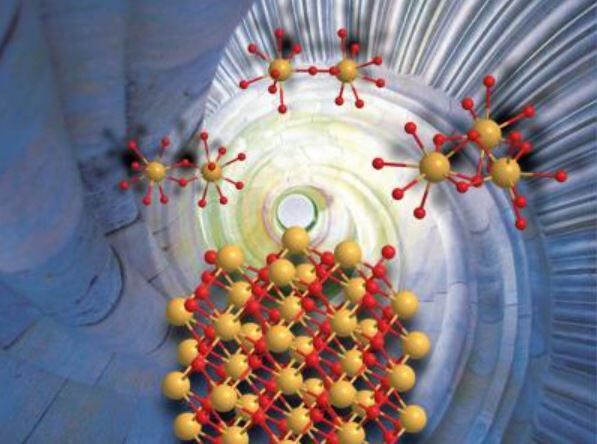
Scientists from the Helmholtz-Zentrum Dresden-Rossendorf (HZDR) and the University of New South Wales have discovered a formation mechanism of nanocrystalline CeO2 particles.
The findings will have a fundamental impact on current methods for producing CeO2 nanocrystals and is likely to simplify the production methods that are currently available. The scientists used a multi-spectroscopic technique to observe the nanocrystal evolution process.
Current methods of synthesising nanocrystalline CeO2 particles are based on sol-gel and thermal treatments or by adding accelerant reagents. The new study has enabled a better understanding of how the metal nanocrystal particles are formed at the atomic scale, in an aqueous solution.
The study found that it is possible to produce uniformly sized nanoparticles of CeO2 in a size suitable for industrial application. The technique involves pH adjustment of tetravalent cerium in an aqueous solution, in the absence of any chemical or heat treatment afterwards.
Dr Christoph Hennig, one of the scientists involved in the study, said that the multi-spectroscopic technique is applicable to other types of research on metal nanocrystals. Nanocrystalline CeO2 particles are used in a variety of industrial and medical applications.
Source: http://www.sciencedaily.com/releases/2013/06/130625121155.htm




 02 9541 3500
02 9541 3500