REDSHIFTBIO AURORA
RedShiftBio Aurora all-in-one, single drop, protein characterisation platform with integrated touchscreen for ultra-sensitive measurements of structural changes.




REDSHIFTBIO AURORA
RedShiftBio Aurora all-in-one, single drop, protein characterisation platform with integrated touchscreen for ultra-sensitive measurements of structural changes.
| Manufacturer | RedShiftBio |
|---|---|
| Product Series | Aurora |
| Measurement principle | Microfluidic Modulation Spectroscopy (MMS) |
| Application | Protein Conformation |
| Sample type | Proteins, Peptides, mAbs, ADCs, AAVs, and mRNA |
| Low sample consumption | 50 µl sample volume |
| Concentration Range | 0.1 mg/mL to >100 mg/mL |
Product Overview
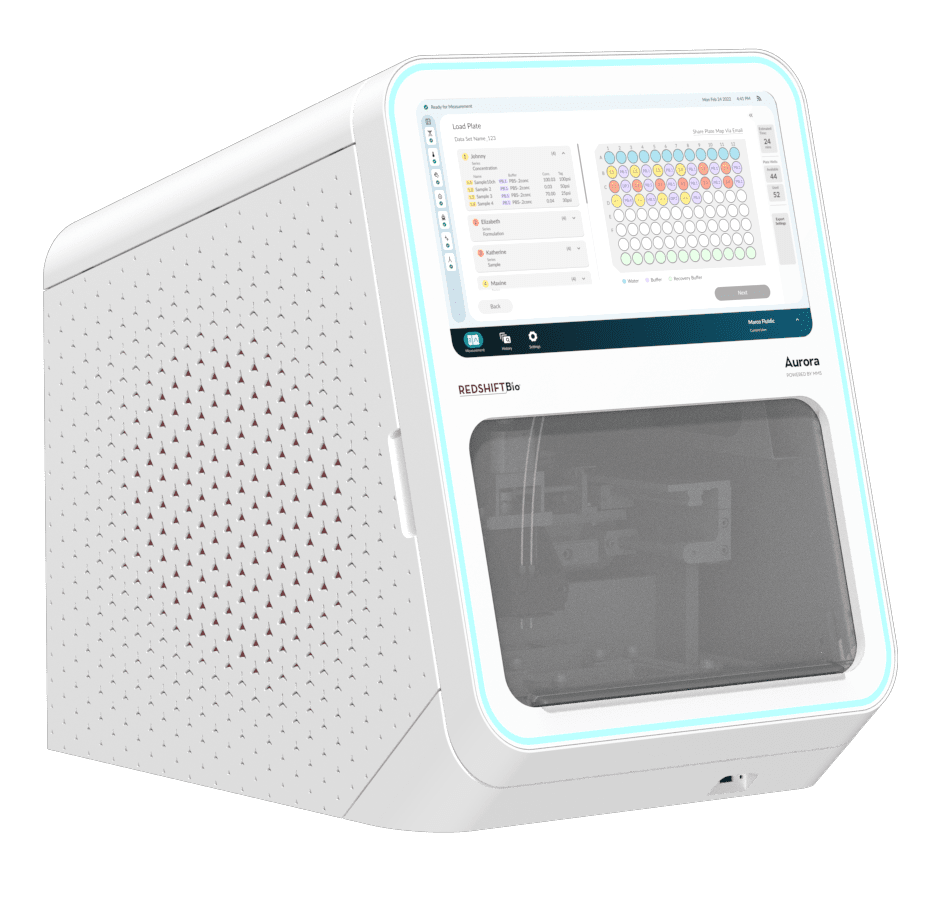
RedShiftBio Aurora provides ultra-sensitive, ultra-precise measurements of the structure of proteins, peptides and other biomolecules through a novel technology called Microfluidic Modulation Spectroscopy (MMS).
Aurora is designed to be a space-saving, all-in-one analytical unit with integrated touchscreen. It offers a 10-fold decrease in sample volume at a lower price point than the first-generation AQS3 Pro and Apollo instruments, making it available to more researchers and applications than ever before. Users can compare higher-order structure and similarity profiles for confidence in structural similarity and activity to inform discovery, screening, formulation, and quality control in biopharmaceutical drug development. It can be used for a wide range of biomolecules from mAb-based biotherapeutics to robust measurements of proteins, enzymes, peptides, ADCs, AAVs, and mRNA.
MMS directly addresses the limitations of traditional spectroscopic methods like CD and FTIR by enabling direct, label-free analysis over a wide concentration range in complex buffer formulations, without the need to buffer exchange or dilute your precious samples.
Contact us for a demo today!
- DEMONSTRATION VIDEO
- BENEFITS
- APPLICATIONS
- CUSTOMER TESTIMONIAL
DEMONSTRATION VIDEO
BENEFITS
- 50 µl sample volume, a dramatic reduction from first generation MMS instrument requirements
- Accurate and reproducible measurements across a broad concentration range from 100 µg/ml to >100 mg/ml, allowing measurements in relevant conditions
- Integrated touchscreen computer interface
- 20x faster and 30x more sensitive to changes in structure than CD or FTIR
- Real-time buffer subtraction minimises background noise and interference from excipients
- Complete hands-free automation for painless data acquisition and analysis
APPLICATIONS
Aggregation – This is a common indicator of protein instability, which can result in a therapeutic product being unfit for launch. MMS is one of the only techniques which can directly monitor the formation of aggregates due to its ability to measure intermolecular beta-sheet structures.
Quantitation of protein concentration – The structure and behavior of proteins in solution can be a function of their concentration, therefore the ability to accurately quantify protein concentration allows better comparison between different proteins and formulations. MMS’s high resolution (<0.001 cm-1) and low stray light susceptibility increases the linear concentration range for the measurement by more than 2 orders of magnitude. With a minimum measurable concentration of less than 10 μg/mL and an upper limit of greater than 200 mg/mL, this technique offers a significant improvement over conventional absorbance-based assays.
Protein Higher Order Structure (HOS) – During manufacturing, biopharmaceutical proteins may undergo conformational changes which can alter their secondary structure and therefore their function. MMS has the ability to detect these changes with great sensitivity, in the formulation buffer and at the concentration of interest, without the need for dilution or deuteration. MMS offers detailed information on which structural motifs in the protein molecule are changing, to help guide the development of more stable protein molecules and formulations.
Protein Stability – Measuring how stable a protein is to thermal or chemical exposure during manufacturing and storage is critical. Structural changes can lead to decreases in potency, degradation of the product, and increases in impurities and aggregates which can be extremely harmful. MMS technology enables the accurate assessment of the stability of the protein throughout the entire formulation, development, and manufacturing process, reducing risks and enabling control strategies for each critical quality attribute (CQA).
Biosimilarity – Along with functional comparisons, measurement and analysis of the structural similarity between proteins is an effective method of demonstrating bioequivalence. MMS measures protein secondary structure, reveals very small conformation differences between different proteins, and provides information as to where those differences occur. These capabilities make MMS a powerful tool in the analysis and development of biosimilars.
POSTER : MMS EVALUATION OF HOS FOR IGG SAMPLES SPIKED WITH BSA
CUSTOMER TESTIMONIAL

Revolutionising protein structural analysis: the RedShiftBio MMS technique
“I was very lucky to have access to MMS technology during my PhD,” Kim said. “It addressed the limitations of traditional protein structural analysis techniques with its accuracy and the ability to measure a wide range of protein concentrations and has enabled the precise characterisation of complex food formulations, particularly those involving plant proteins.” Read full article here https://bit.ly/45jmoJq
Product Enquiry
SPEAK TO OUR AUSTRALIAN AND NEW ZEALAND TEAM
If you would like to enquire about this product, fill out this form and one of our product specialists will contact you shortly.
If you need help in the operation or repair of an instrument then please contact the Customer Help Centre directly.
Please note, product enquiries are only applicable to Australia, New Zealand, Papua New Guinea and Fiji
Or call us instead at (02) 9541 3500




 02 9541 3500
02 9541 3500

